A tap is opened to fill a bucket, and water flows from the tank through a pipe. Now, as shown in the diagram, consider two points A and B on the vertical part of the pipe which has a uniform cross section.
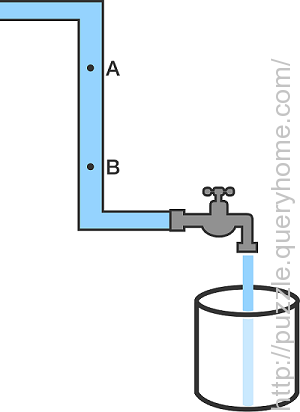
If the pipe is completely filled with water and the water flows steadily, then at which point is the speed of the water flow greater?
